Current PSA screening recommendations take into account your age, ethnicity and family history. Want to know what role your underlying genetic risk plays?

● Understand your risk of prostate cancer
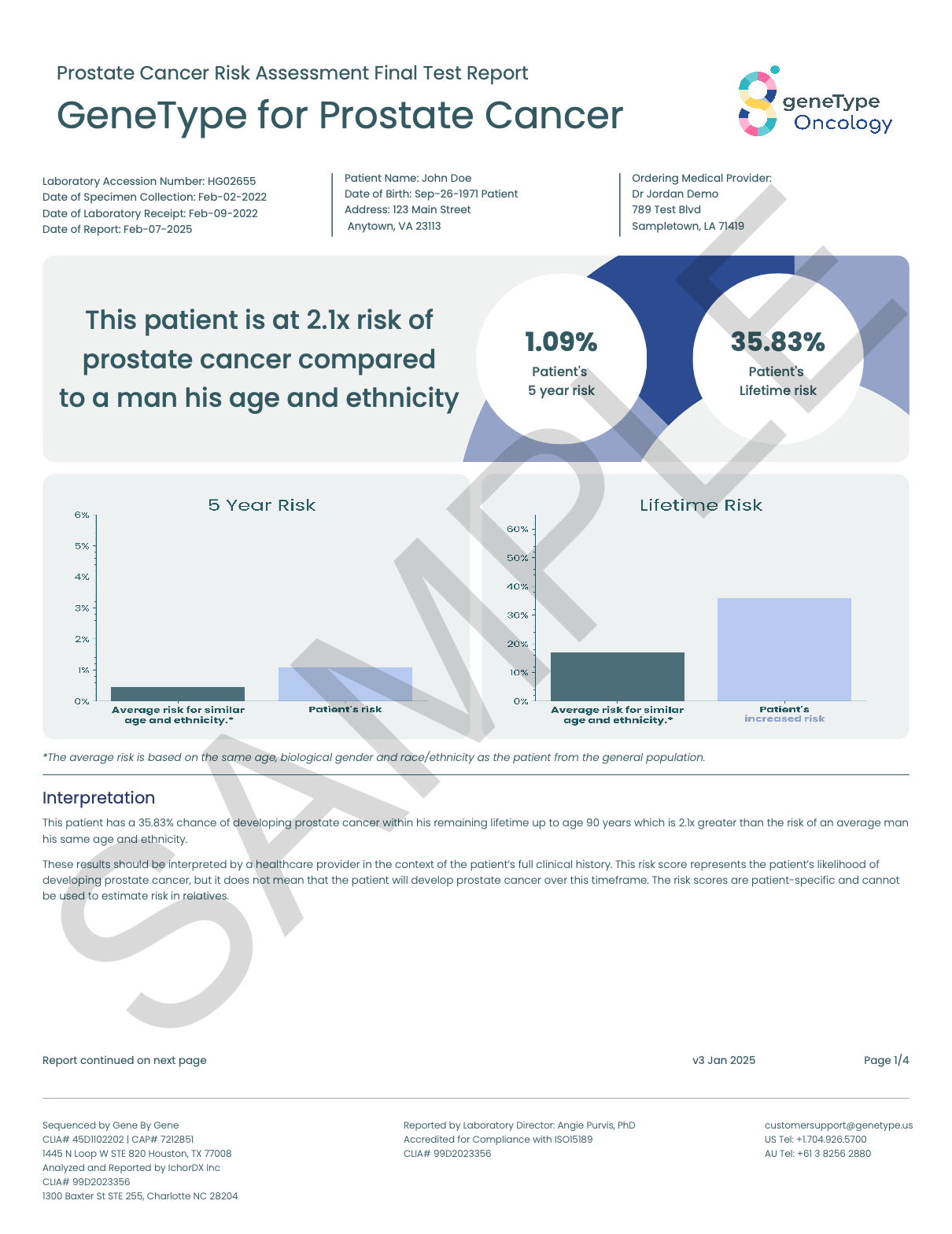
● A lifetime risk score and a five-year risk score
● Discussion points around screening recommendations from the NCCN and USPSTF

Would you benefit from a PSA screening before the age of 55?
If you are over 55, how are you making the decision to undergo periodic PSA screening?
geneType™ can help you have a joint-decision-making discussion with your HCP.


Get a clearer picture of your risk.
Make screening decisions that fit your needs.
Know your risk of prostate cancer, so you can take action.

Register for your geneType™ test using our portal.
Depending on the test, you may require access to your medical records (ex. lipid panel or breast density results).
Payment will be collected at time of registration.

Your clinical information will be reviewed by our third party telehealth partner, DNA Visit.
If you qualify for testing, a kit will be shipped to your address on file.
If you do not qualify for testing, you will receive a refund.

Complete buccal sample collection as instructed and return in pre-paid envelope.
Do not eat, drink, smoke, vape or chew gum within 30 minutes of collection.

When your results are ready, you will have the chance to speak with your DNA Visit healthcare provider.
All at-risk patients will be required to speak with the provider prior to results-release.
If your question is not shown here, please Contact us directly.
No. At this time geneType™ is a self-pay test. However, you may use FSA/HSA to pay for the test.
This test is not applicable to men who have personal history of prostate cancer or who have already been shown to have a cancer mutation, for example in the BRCA1 or BRCA2 gene, or a diagnosis of a genetic syndrome that may be associated with an elevated risk of prostate cancer.
Cancer is a multifactorial disease and there are many different risk factors. Of these, geneType™ integrates the most predictive risk factors into the model. Future models may include additional risk factors as the scientific community’s understanding of prostate cancer continues to evolve.

*Patient eligibility dependent on personal medical history, age and sex
The Multi-Risk suite of tests is for adults 40-85 years of age. At maximum, a woman would be eligible for eight diseases in the panel; a man would be eligible for seven. Starting at age 30, a patient may qualify for geneType’s™ cancer risk assessments only.
1.American Cancer Society. Prostate Cancer Screening Recommendations. Available at: https://www.cancer.org/cancer/prostate-cancer/detection-diagnosisstaging/acs-recommendations.html. Accessed June 2022.
2. Fenton JJ et al. JAMA. 2018 ;319(18):1914-1931.
3. NCCN Clinical Practice Guidelines in Oncology: Prostate Cancer Early Detection. V1.2022; Feb 2022. Accessed July 2022

Accredited For Compliance With NPAAC Standards And ISO 15189