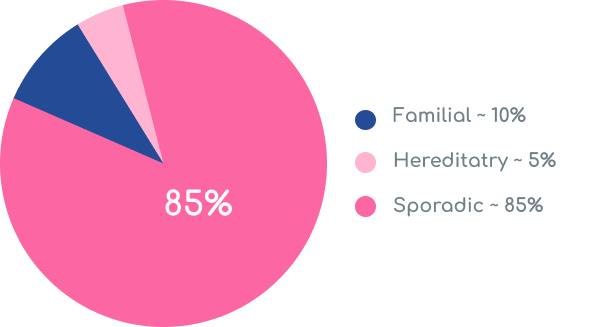
Sporadic disease is the most commonly diagnosed form of breast cancer, meaning these women don’t have significant family history nor are they pathogenic variant carriers at diagnosis!
The value of an integrated risk assessment
One in 200 women are positive for a hereditary breast cancer associated pathogenic variant.
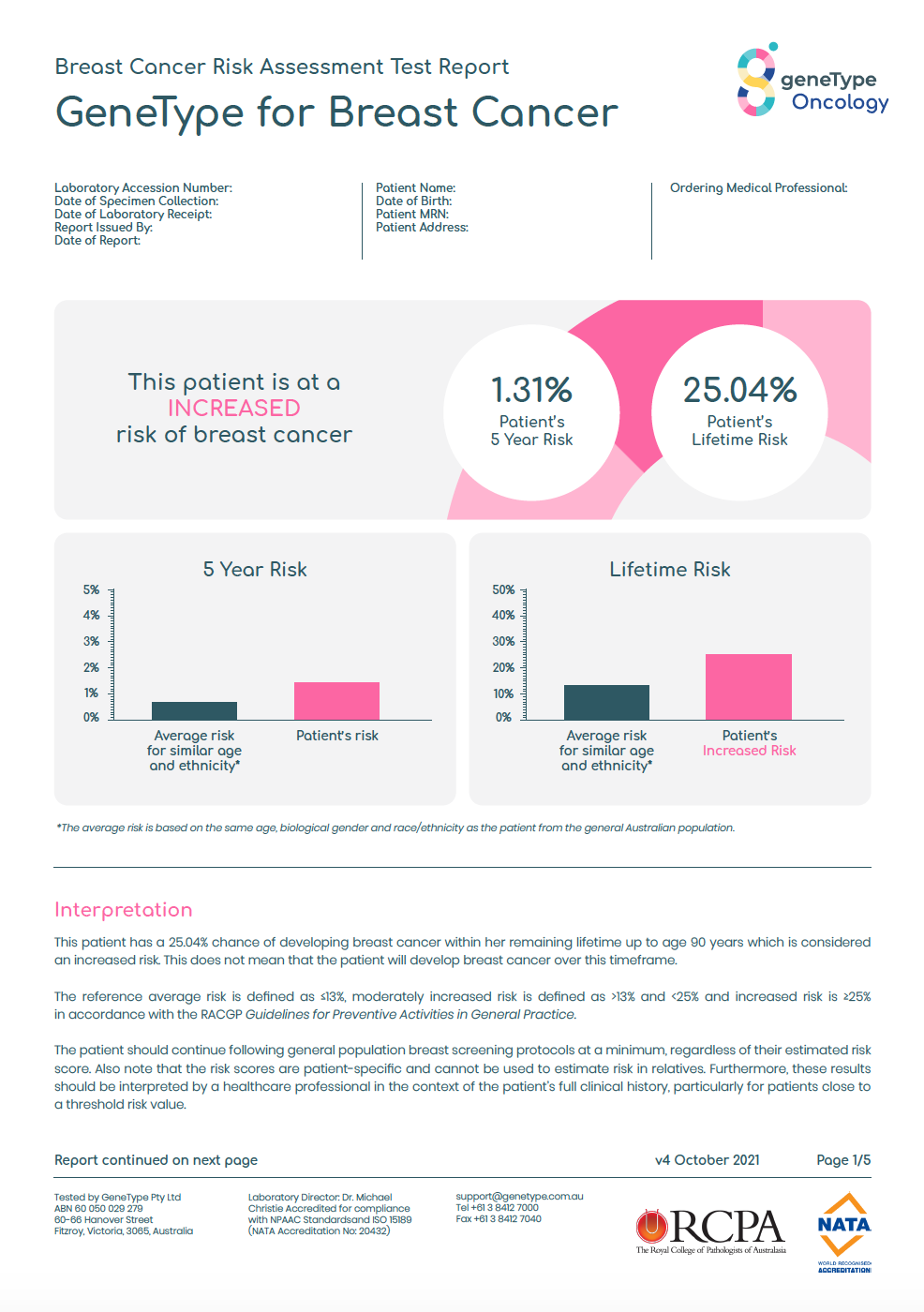
One in 5 women will have a combination of risk factors that put them at an actionable, increased risk.*
*using established clinical thresholds 5-year risk threshold of 1.67% or 3%; lifetime risk threshold of 20%